Site Search
Displaying results 71 - 80 of 573

Resource | Publications,
Each year, the AVAC Report frames the most pressing advocacy issues facing the HIV response. At the threshold of 2020, it’s clear that global goals for HIV prevention will miss the mark by a long shot. Though important progress has been made, the crisis UNAIDS called out in 2016 persists today with new infections around 1.7 million annually, a far cry from the 2020 target of fewer than 500,000. So, we asked ourselves, Now What?, and answered with cross-cutting analysis and an advocacy agenda to match.

Resource | Publications,
These guidelines provide further reassurance of DTG as the preferred antiretroviral (ARV) drug in first- and second-line regimens due to the declining estimate of neural tube defect risk and observed efficacy. This reassurance comes at a time when pretreatment resistance to non-nucleoside reverse-transcriptase inhibitors (NNRTI) is increasing in low- and middle-income countries, creating demand for access to alternative non-NNRTI ARV drugs.

Resource | Fact Sheets,
In Hong Kong, the estimated HCV prevalence in general population is around 0.3%. In the last decade, there have been increasing overseas reports of acute HCV infections via sexual transmission in men who have sex with men (MSM), especially those who were HIV positive. Locally, the prevalence of HCV in the HIV infected MSM seen at the Department of Health (DH) was found to be 1.3% for the years 2000-2012, which is 4 times that of the general population. In addition, rising trend of the infection among local HIV infected MSM has been observed in recent years. In 2013, a case series of sexually transmitted HCV infections among HIV infected MSM was found by DH.

Resource | Publications,
This edition of UNICEF’s report on requirements for humanitarian action highlights major emergencies affecting children and families around the world, and the results achieved by UNICEF and partners in response to those crises. Noting that more violent conflicts are raging today than at any time since the adoption of the Convention on the Rights of the Child 30 years ago, the report also describes UNICEF initiatives to improve the quality of its humanitarian response in 2019 – particularly in high-threat contexts.

Resource | Publications,
By working with communities, uniformed services, governments and regional and international partners, UNAIDS supports the 2,57 million people living with HIV affected by humanitarian disasters globally to ensure that they have access to programmes preventing and addressing gender-based violence, and the HIV services they need.

Resource | Publications,
Viet Nam is facing a rapidly growing HIV epidemic that is beginning to extend beyond initial concentrations in networks of injecting drug users and sex workers. The number of people living with HIV doubled between 2000 and 2005, from approximately 122,000 to 263,000. The adult HIV prevalence is estimated to be 0.5% at the national level in 2005 but exceeded 1% in several provinces. There were an estimated 37,000 new infections in 2005. Due to increased heterosexual transmission, the number of infected females compared with males is increasing each year. In 2005, the ratio was estimated to be 2 to 1, males to females. The number of AIDS-related deaths is growing and is estimated to have increased from 9000 in 2003 to 14,000 in 2005.

Resource | Publications,
The first case of HIV infection in Papua New Guinea was detected in 1987. By June 2005, 12,341 people had been reported to be living with HIV/AIDS. The country is facing a generalized epidemic with rapidly increasing prevalence in a difficult socioeconomic context. A national epidemiological consensus meeting in November 2004 estimated an average prevalence rate of 1.7%, and between 25,000 and 69,000 people with 15-49 years were living with HIV/AIDS. Prevalence rates among women attending antenatal care services are estimated to vary between 1% and 4%. Available data suggests that the epidemic is predominantly transmitted through heterosexual contact (84%), fuelled by high-risk behaviour including widespread commercial and casual sex. Approximately 93.1% of current reported cases are adults.
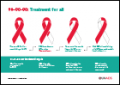
Resource | Infographics,
There are 37.9 million people living with HIV. 79% know they are HIV-positive. The rest do not. Three out of five people living with HIV are on antiretroviral therapy. Only 53% of people living with HIV have undetectable levels of the virus.

Resource | Publications,
This document summarizes the proceedings of the Regional HIV Forum for the Pacific Island Countries and Territories 2018. The forum was the first event of its kind in the Western Pacific, bringing together people living with HIV and health care workers from across the region to meet, discuss and share their experiences working with and living with HIV. It provided a platform for open dialogue for both groups, enhancing HIV and antiretroviral therapy literacy, and self-empowerment, while aiming to catalyse coordination across HIV care, treatment and prevention programmes in the Pacific islands.

Resource | Publications,
The primary objective of the further analysis of the 2016 NDHS is to provide more in-depth knowledge and insights into key issues that emerged from the survey. This information provides guidance for planning, implementing, refocusing, monitoring, and evaluating health programs in Nepal. The longterm objective of the further analysis is to strengthen the technical capacity of local institutions and individuals for analyzing and using data from complex national population and health surveys to better understand specific issues related to country need.





